Technology could improve results, allow non-burn surgeons to respond to mass casualty emergency
Date: September 19, 2013
Organization: Spectral MD Inc., of Dallas, Texas
Project funding: $2.4 million under a 15 month contract which can be extended for a total of up to five years and up to $13.1 million
| About this project: Under the BARDA program, Spectral MD will continue developing its DeepView imaging system. The system analyzes blood flow to identify the boundaries of dead, damaged and healthy tissue. In order to increase the likely success of skin grafting, dead or damaged skin is removed without removing any healthy skin tissue.
Use of such a device could improve the healthcare system’s response to burn patients in a radiological or nuclear emergency, such as the detonation of a nuclear device.
Today, experienced surgeons visually inspect burned wounds to find where dead skin stops and live skin tissue begins. This system is capable of scanning large portions of a patient’s body, performing imaging prior to removal of dead and grafting healthy skin anywhere on the patient’s body. |
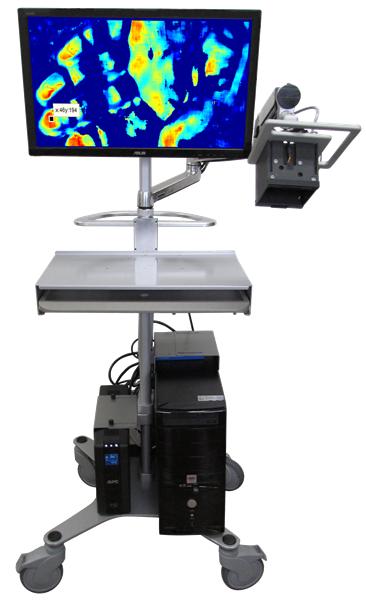
|
Finding the precise points at which dead skin ends and live skin tissue begins, helps surgeons reduce the amount of the grafting products needed in a mass casualty situation and could improve surgical outcomes for patients, possibly saving lives and shortening their hospital stays.
The detonation of a nuclear bomb or improvised nuclear device produces intense heat, potentially resulting in severe burns that can cover large portions of a patient’s body. Such thermal burns typically require specialized treatment, including surgery for skin grafts. In a radiological or nuclear emergency, surgeons could have thousands of patients in need of immediate skin grafting to save their lives.
A device that makes the distinction between dead and live skin could enable doctors without extensive skin grafting experience to respond to the disaster.
If the contract is extended, the company would conduct clinical trials to demonstrate safety and efficiency. The device also may have commercial applications for other deep tissue injuries such as treating diabetic skin ulcers.
Many of the products that BARDA is developing may find uses outside of emergency medicine, especially in the case of severe thermal burns. The development of such medical products improves the nation’s preparedness by increasing the number of products available for emergencies. With multiple uses the market for these emergency products can be sustained because the products also can be used in day-to-day medical situations.
